Eli Lilly has released new late stage results for its next major obesity drug candidate, retatrutide, and the numbers are turning heads across the medical and investing world. The company says the treatment produced the most significant weight loss seen in any late stage study to date, while also sharply reducing knee osteoarthritis pain. The findings clear one of several major hurdles ahead as Lilly pushes to cement its leadership in the booming global obesity drug market.
The data adds momentum to a drug pipeline that has already reshaped Lilly into one of the world’s most valuable pharmaceutical companies. Investors immediately reacted, sending shares up more than 3 percent on Thursday.
Big Weight Loss Results That Rival Bariatric Surgery
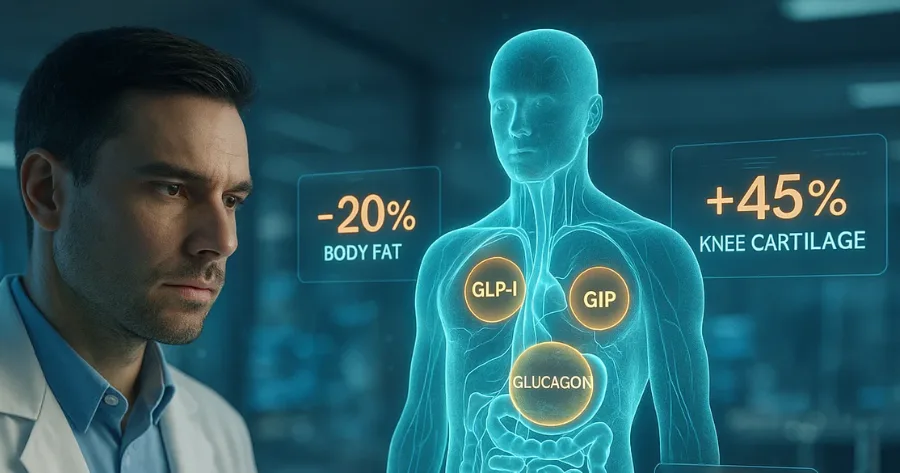
Retatrutide delivered substantial weight loss for adults with obesity and knee osteoarthritis. On the highest dose, patients lost an average of 23.7 percent of their body weight at 68 weeks when including all participants, even those who stopped treatment early. Among patients who remained on the drug for the full duration of the study, average weight loss reached 28.7 percent.
Doctors involved with the research say the magnitude of weight loss is historic. Dr. Caroline Apovian of Brigham and Women’s Hospital stated, “It’s incredible. Now, we have a drug that rivals the weight loss benefits of surgery.”
Some participants reportedly dropped so much weight so quickly that they chose to withdraw from the study.
This level of effectiveness suggests Lilly is positioning retatrutide for people with severe obesity, defined as a body mass index above 35 or 40. According to the company, 84 percent of people in the trial fell into this category.
A New Approach to Treating Obesity
Retatrutide is the first of Lilly’s next generation obesity drugs to reach a phase three trial. It functions differently from current market leaders and appears to have stronger physiological effects on appetite and satiety. Retatrutide activates three hormonal pathways at once: GLP 1, GIP and glucagon. For this reason, researchers call it a triple G drug.
By comparison, Zepbound and other tirzepatide based treatments activate only GLP 1 and GIP. Novo Nordisk’s Wegovy activates just GLP 1. The triple approach may explain why retatrutide produced more potent weight loss than existing drugs.
For investors, this is part of Lilly’s strategy to maintain dominance in a market that analysts estimate could reach 100 billion dollars by the 2030s. Retatrutide is expected to become the next pillar of Lilly’s obesity franchise alongside Zepbound and its upcoming oral weight loss pill.
Knee Pain Relief Adds a New Value Proposition
While weight reduction was a major focus of media coverage, one of the trial’s primary endpoints involved relief of knee osteoarthritis pain. On average, retatrutide reduced knee pain by up to 62.6 percent based on a widely used survey of patient symptoms. More than one in eight participants ended the trial entirely pain free.
Osteoarthritis impacts tens of millions of adults, and obesity is a major contributing factor. A treatment that simultaneously reduces weight and pain could offer a stronger therapeutic benefit than current injectable options.
Apovian noted that current weight loss drugs sometimes lead to lean muscle loss, raising concerns about mobility. She said the physical function improvements seen in this trial indicate that retatrutide may address that concern for people with severe obesity.
Side Effects and Dropout Rates
While the weight loss results exceeded Wall Street expectations, tolerability remains an important competitive factor. JPMorgan analyst Chris Schott said retatrutide’s tolerability data is “somewhat worse vs Zepbound, though not surprising, in our view.”
Among patients on the highest dose, 18 percent discontinued treatment because of side effects. The placebo group saw a discontinuation rate of 4 percent. Eli Lilly said dropout rates were “highly correlated” with starting BMI levels and in some cases were linked to “perceived excessive weight loss.”
For comparison, among patients with a BMI of 35 or higher who used the highest dose, 12 percent discontinued treatment. Schott noted that this aligns more closely with dropout rates observed for Zepbound and Novo Nordisk’s Wegovy.
The most common side effects were gastrointestinal, including nausea in 43 percent of patients, diarrhea in roughly 33 percent and vomiting in 20.9 percent. More than 20 percent experienced dysesthesia, described as an unpleasant nerve related sensation, although Lilly stated it was generally mild and rarely caused participants to withdraw.
Analysts Expected Strong Results, but Retatrutide Surpassed Them
Ahead of the data release, BMO Capital Markets analyst Evan Seigerman anticipated weight loss in the 20 to 23 percent range with at least a 50 percent reduction in knee pain. The actual results exceeded both benchmarks. Seigerman later wrote that while discontinuation rates were notable, “all in results are impressive.”
Kenneth Custer, president of Lilly Cardiometabolic Health, said the company believes retatrutide “could become an important option for patients with significant weight loss needs and certain complications, including knee osteoarthritis.”
The Competitive Landscape: Novo Nordisk Racing to Catch Up
Novo Nordisk is not sitting still. The company recently agreed to pay up to 2 billion dollars for rights to an early stage experimental drug developed by United Laboratories International in China. Like retatrutide, Novo Nordisk’s new asset uses a three hormone approach and could become a direct competitor. However, it remains in early development and is likely years away from commercialization.
Meanwhile, Zepbound continues to deliver strong real world performance, with higher dose tirzepatide producing roughly 20.9 percent weight loss on average in earlier late stage studies.
Even as competitors race to match these outcomes, Lilly’s head start with triple agonist therapies puts it in a strong strategic position.
What Comes Next: Seven More Trials on the Horizon
The TRIUMPH 4 study did not focus solely on weight loss. Lilly has seven more late stage trials designed specifically to measure different aspects of retatrutide’s effectiveness, safety and durability. The company expects to release results from these studies through 2026. If performance remains consistent, approval and commercial launch could transform the already explosive obesity drug market.
For investors, retail and institutional interest in obesity related pharmaceutical stocks remains high. Lilly and Novo Nordisk have become two of the most influential market movers of the past 18 months as demand for these drugs outpaces supply and drives new revenue projections across the sector.
Why This Matters for Investors
Obesity drugs are no longer simply another therapeutic category. They have become a defining force in global health care markets and a major contributor to GDP level productivity gains. Effective treatments lead to fewer chronic diseases, lower health care costs and greater workforce participation.
Retatrutide’s strong trial results reinforce the idea that obesity treatments are entering a second wave of innovation. Drugs are becoming more powerful, more targeted and more versatile. A treatment that addresses severe obesity and joint pain at the same time has the potential to widen the patient population and extend insurance coverage.
If future data confirms retatrutide’s advantages, Eli Lilly could secure an even larger share of what many analysts now call the most important pharmaceutical market of the decade.